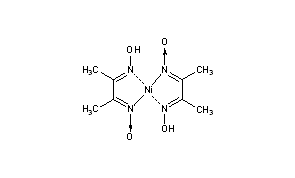
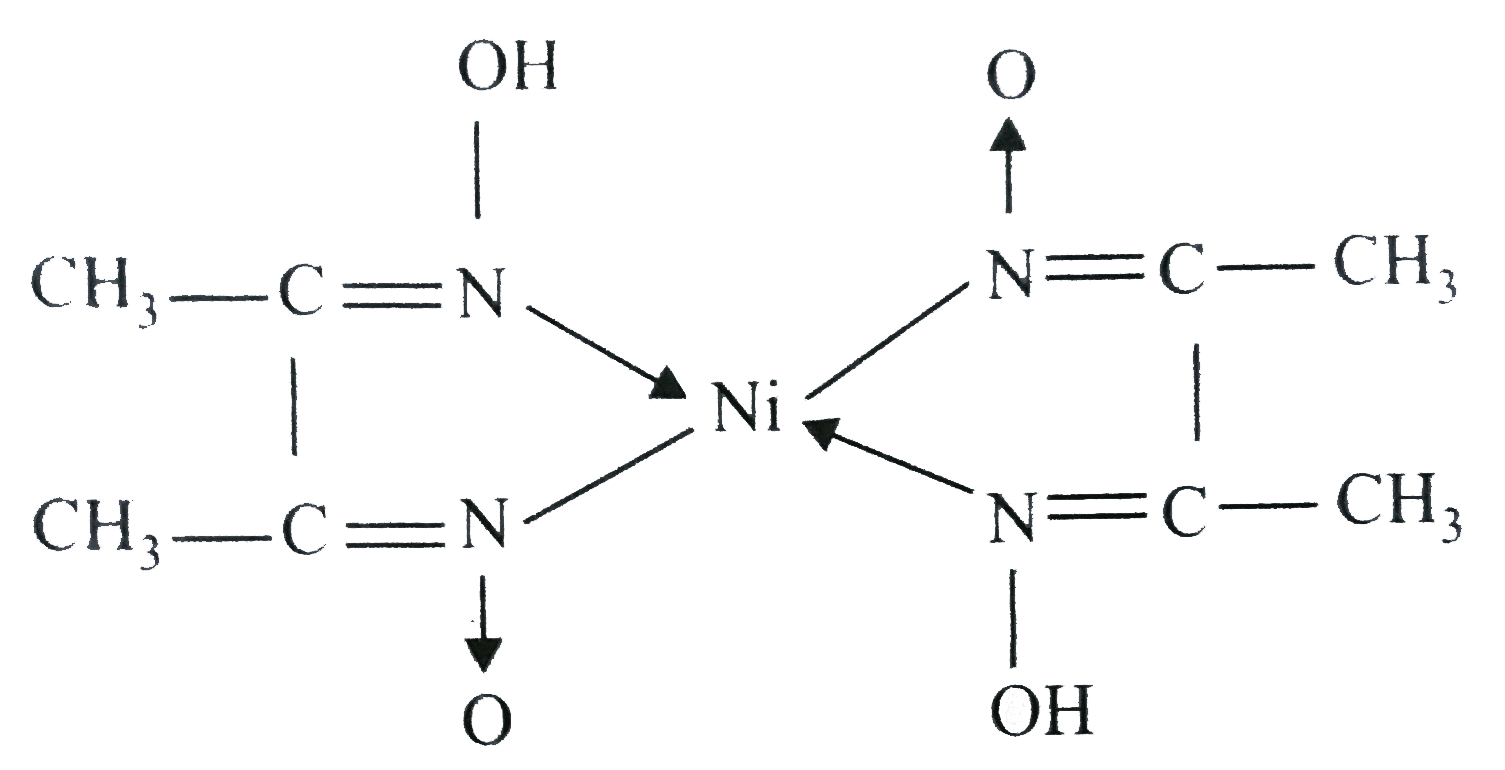
Dimethylglyoxime (C4H8N2O2) - Structure, Molecular Mass, Properties and Uses of Dimethylglyoxime, Dimethylglyoximato Ligand

In basic medium the amount of Ni2+ in a solution can be estimated with the dimethylglyoxime reagent. The correct statement(s) about the reaction and the product is(are):
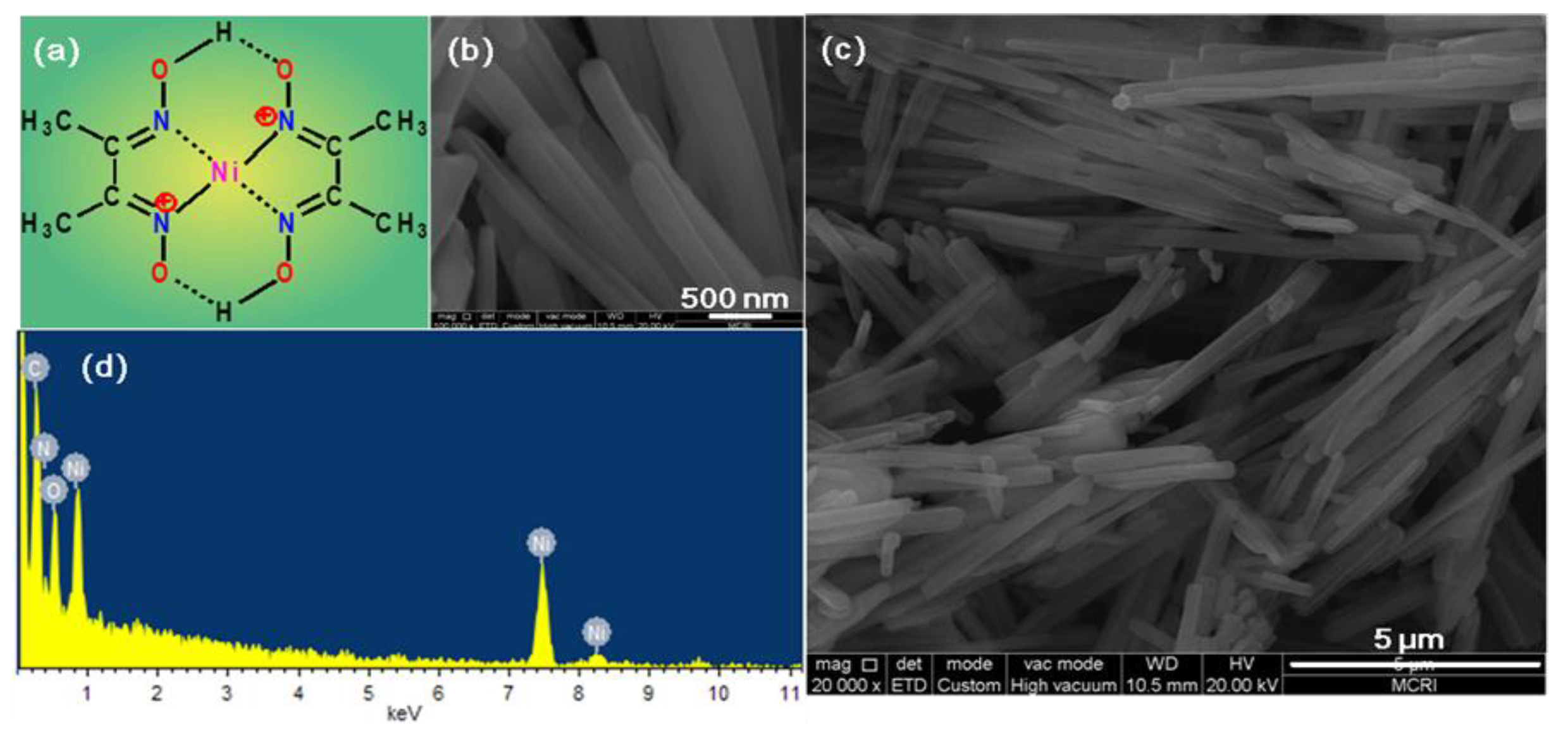
Catalysts | Free Full-Text | Study on Thermal Decomposition Behavior, Gaseous Products, and Kinetic Analysis of Bis-(Dimethylglyoximato) Nickel(II) Complex Using TG-DSC-FTIR-MS Technique
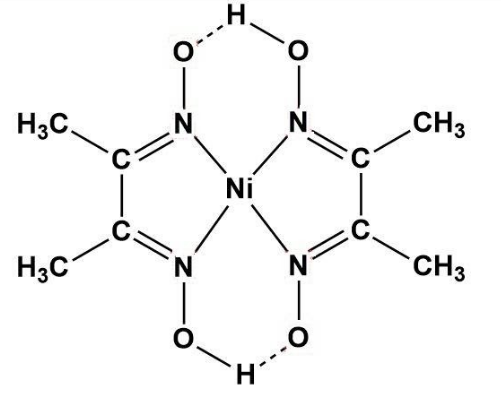
An alcoholic solution of dimethylglyoxime is added to an aqueous solution of nickel (II) chloride. Slow addition of ammonium hydroxide leads to the precipitation of a rosy red coloured metal complex. Then